CellCrine
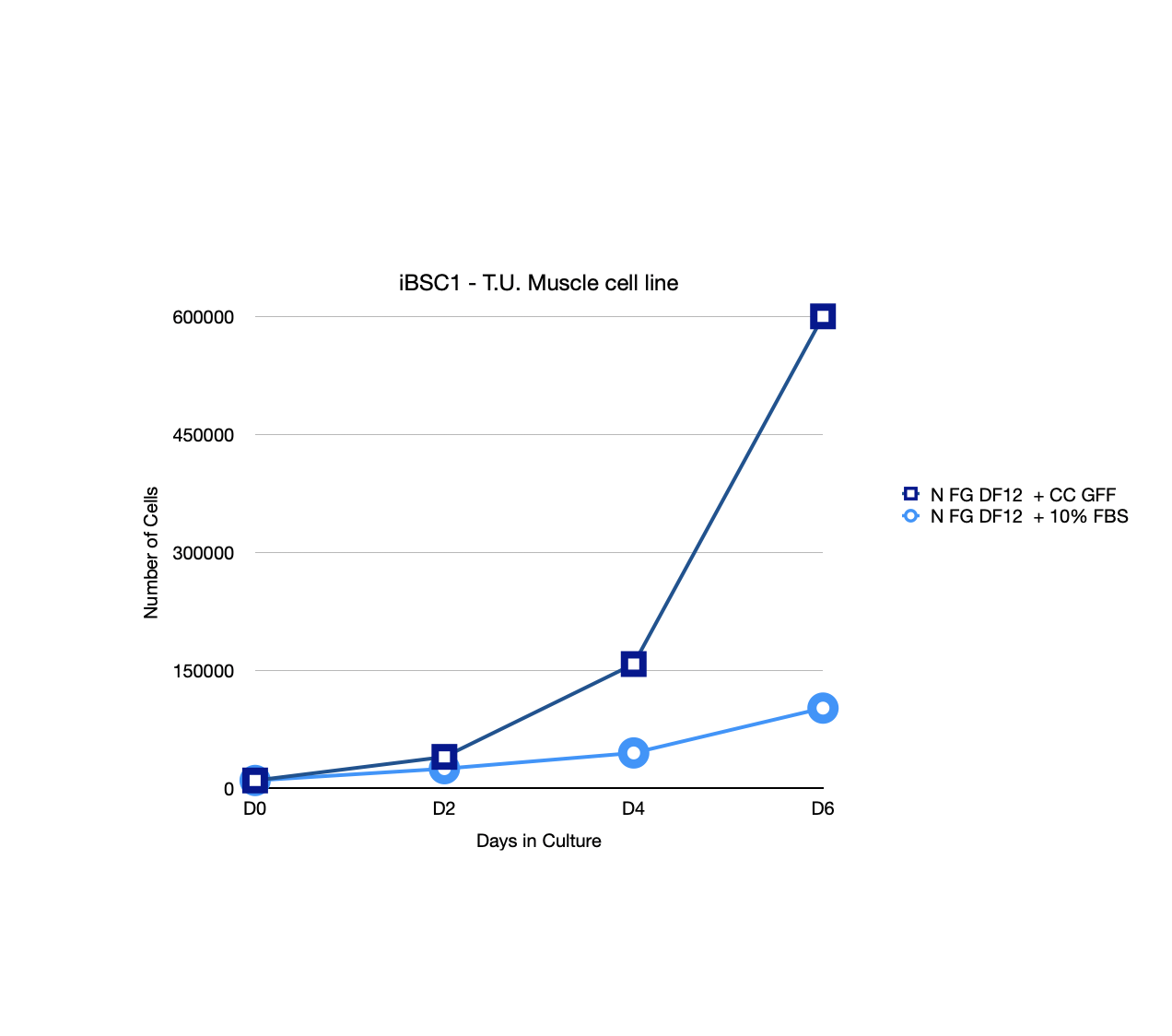
A youthful protein* and an optimal Basal media outperforms FBS across all cell types tested to date
* Identified, Isolated, and sequenced. Effective in all cell types tested to date.
We predict that it will work for all cell types.
CellCrine’s Approach.
We have engineered the media to fit the cell
Unlike GMO-based methods that focus on engineering cells to fit existing media, CellCrine utilizes a non-GMO approach to engineer the media to fit the specific needs of the cell. This youthful protein has proven effective in all cell types tested so far, and we predict it will work for virtually all cell types.
Patent Pending CC Tech
We have developed a groundbreaking BAP Chemically Defined Supplement (BAP-CDS) that outperforms Fetal Bovine Serum (FBS).
CellCrine’s mission is to apply and validate BAP CDS across all cell types including cell lines and primary cells.
The Chemically Defined Media (CDM) market is currently poised for significant growth, with projections expected to exceed $4 billion by 2026.
As the biopharmaceutical, cell therapy, and regenerative medicine sectors continue to expand, the demand for high-quality, consistent, and scalable culture media has become increasingly critical to emerging trends in laboratory automation, and sustainability. Given its foundational role in bioprocessing, the CDM market remains a strategic focus for investors and corporations.
By identifying and optimizing critical media components that induce potent cell signaling, we have successfully utilized BAP-CDS to eliminate the need for traditional growth factors.
BAP-CDS has been validated ( primary and cell lines ) on more than 20 different cell types, including MSCs, Skin cells, Tuna, CHO, MDCK, MRC5, HeLa, HEK293, Bovine, Porcine, Chicken, Stem cells, Vero, CEF/DF1, and iBSC.
Cracking the code
CellCrine is proud to announce a significant milestone following 30 years of dedicated research and development. We have developed a groundbreaking BAP Chemically Defined Supplement (BAP-CDS) technology that consistently outperforms Fetal Bovine Serum (FBS).
Our BAP-CDS allows cells to be seeded in scaffolds and standard cell culture-grade vessels, providing the essential nutrients and building blocks needed to thrive and exceed the performance of FBS.
Cultivated meat companies currently face the challenge of entering cost-sensitive markets where consumer loyalty to traditional animal products remains high. To succeed, these companies require a scaling strategy that prioritizes long-term profitability through competitive pricing and superior taste. CellCrine developed BAP-CDS specifically to address these hurdles and make cultivated meat a market reality.
Our goal is to provide the most cost-effective CDM to the CMI, BioPharma, and scientific communities.
For more information, please contact us at: info@cellcrine.com.
